With over 200 dedicated professionals, Beijing East IP has helped a full spectrum of clients – from startups to Fortune 500 corporations to domestic multinational companies – on their intellectual property issues in China.
Admissibility of Late-filed Experimental Data is the Matter in China
Author:Jian LI
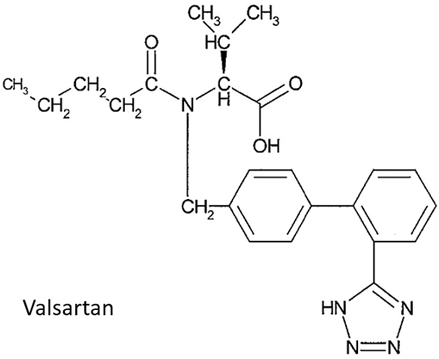
Just at the beginning of this year, the Patent Reexamination Board (PRB) of SIPO issued a decision in a notable patent invalidation trial against Novartis’s patent on Entresto (Sacubitril/Valsartan), holding all the claims of the patent invalid as being obvious.
The patent at issue is CN Patent No. 201110029600.7 (the “’600 patent”) entitled Pharmaceutical Composition Comprising Valsartan and NEP Inhibitor, claiming the priority of U.S. Provisional Appl. No. 60/349,660, and issued to Novartis on April 8, 2015. It claims a pharmaceutical composition comprising: (i) the AT 1-antagonist Valsartan or a pharmaceutically acceptable salt thereof; (ii) N-(3-carboxy-1-oxopropyl)- (4S)-(p-phenylphenylmethyl)-4-amino-2R-methylbutanoic acid ethyl ester or N-(3-carboxy-1-oxopropyl)-(4S)- (p-phenylphenylmethyl) -4-amino -2R-methylbutanoic butyl ester or a pharmaceutically acceptable salt thereof; and a pharmaceutically acceptable carrier. The claimed combination of Valsartan and the NEP inhibitor has been found to achieve greater therapeutic effect than the administration of Valsartan, ACE inhibitors or NEP inhibitors alone and promotes less angioedema than is seen with the administration of a vasopeptidase inhibitor alone.
The patented formulation was approved by USFDA in July, 2015 and by CFDA in July, 2017 as a new drug for the treatment of heart failure. Novartis marketed the drug under the brand name Entresto, which has been evaluated worth 10 billion USD, as a blockbuster for regaining its market share of cardiovascular drugs.
As disclosed by Center for Drug Evaluation of CFDA, for Sacubitril/Valsartan at least two domestic generic companies have filed an application for clinic trial for new chemical drugs under Class 2.1 up to now. And it is not a bit surprising that a request for invalidation of the ’600 patent was filed in April, 2017 by someone who seems to be a straw man.
The grounds for invalidation raised in the request are as follows: i) the enablement requirement is not met; ii) the claims are not supported by the description; iii) the claim scope is indefinite; and iv) the claimed invention does not have the inventiveness. The PRB did not accept the grounds i) – iii), but found all the claims of the ’600 patent obvious in view of the prior art, thus declaring the patent invalid.
The key issue in the PRB’s negative decision on the patent’s inventiveness relates to the admissibility of late-filed supplemental experimental data as evidence for evaluating the inventiveness.
The PRB found that a combination of some AT 1-antagonists with some NEP inhibitors as a treatment for hypertension and congestive heart failure was broadly disclosed by the prior art. Claim 1 of the ’600 patent is drawn to a combination of the specific AT 1-antagonist Valsartan and the specific NEP inhibitor N-(3-carboxy-1-oxopropyl)-(4S)-(p-phenylphenylmethyl)-4-amino-2R- methylbutanoic acid ethyl ester or N-(3-carboxy-1-oxopropyl)-(4S)- (p-phenylphenylmethyl) -4-amino -2R-methylbutanoic acid butyl ester. Thus it is the technical effect the pharmaceutical composition of claim 1 can achieve in lowering blood pressure that is critical for establishing the inventiveness.
The PRB held that a drug combination should be a well-known principle for selecting drugs for hypertension, as it is possible to reduce the dosage of each of the drugs and thus the side effects thereof, and even to achieve some synergic effects. Such synergic effects can result from, however, only a few specific combinations, instead of any arbitrary one, among numerous possibilities. Therefore, whether the specific combination of claim 1 can really achieve the alleged synergic effects should be verified by sufficient probative experimental results of efficiency test. However, no such concrete experimental data or results were provided in the original description of the ’600 patent that could demonstrate any synergic effect or unexpected therapeutic effect of the claimed invention.
Novartis had submitted supplemental experimental data during the invalidation proceeding to demonstrate the synergic effect of the claimed pharmaceutical combination that lowers the mean arterial pressure in some hypertensive animal models. However, the PRB held that the late-filed experimental data was inadmissible for the following reasons: i) the technical effect to be verified was not mentioned in the original description; and ii) the DOCA-salt and SHR rat models were used in the original description, while the supplemental experiments were conducted on the Dah1, SHPsp, and SHR rat models, with the pharmaceutical combination showing no synergic effect in the SHR rat model, which was originally disclosed. Without admitting the late-filed experimental data as evidence for proving the synergic effect, the PRB denied the inventiveness of the claimed pharmaceutical combination.
The PRB’s holding on inadmissibility of the post-filing experimental data actually follows the Supreme Court’s opinion in the precedents such as Takeda Pharmaceutical Company v. PRB((2012)知行字第41号) that late-filed experimental data can possibly be considered as admissible evidence only when the technical effect to be demonstrated has already been mentioned in the original description, and the experimental data shall be obtainable by a person skilled in the art from the disclosure of the original description.
The present case reaffirms that in China the experimental data play a critical role in the validity of patents related to the chemical and biological technologies. China applies the so-called problem-solution-effect approach in determining the essential aspects of patent validity, such as whether a claimed invention possesses the inventiveness over the prior art, whether the original description contains statutorily sufficient disclosure, and whether a claimed invention is an eligible subject matter for patent. When applying the problem-solution-effect approach, such three questions are always asked as what technical problem is intended to be solved, what technical solution is used to solve the problem, and what technical effect is achieved. The answers to these questions will be taken as the basic and indispensable factual findings used for resolving e.g., the inventiveness and the sufficient disclosure issues. Depending on the nature of an invention, experimental data are required to verify the technical effect the invention can achieve. For example, for an invention on a pharmaceutical compound or composition, experimental results or data are required to be provided in the original description so as to convince a person skilled in the art that the pharmaceutical compound or composition can achieve the intended therapeutic effect for treating a target disease on the presumption that the achievability of such therapeutic effect cannot be inferred from the structure or components of a pharmaceutical compound or composition. Failure to disclose the experimental results or data in the description will incur criticism for insufficient disclosure, leading to rejection of a patent application or invalidation of an issued patent. For some inventions on a composition of matter e.g., a combination of the specific AT 1-antagonist Valsartan and the specific NEP inhibitor as claimed by the’600 patent, some synergic effects the composition can achieve in solving a technical problem, e.g., treating a target disease will have a great weight in determining the inventiveness of the invention. Arguably unexpected, however, the synergic effect need to be demonstrated by sufficient probative experimental data or results. With no such experimental data or results provided, nowhere goes any argument for the inventiveness relying on the synergic or unexpected effect.
Moreover, China takes a first-to-file patent system. Under the first-to-file patent system, basically only the disclosure contained in the original description as filed on the filing date can serve as the factual basis on which the inventiveness and enablement can be judged, while any experimental data submitted after the filing date are not a portion of the disclosure of the original description. It is believed that the public interests must be compromised if little restriction is imposed upon admissibility of the late-filed experimental data.
In the 2010 version of Guidelines for Patent Examination, it is clearly provided that any experimental data submitted after the filing date shall not be taken into consideration for judging whether or not the description meets the requirement of sufficient disclosure. Examiners at SIPO were apt to rigidly apply this provision by categorically refusing any late-filed experimental data submitted for any purpose. This harsh way of dealing with late-filed experimental data was extensively controversial, giving rise to many complaints from applicants and patentees, especially those from other jurisdictions where more flexible criteria on admissibility of late-filed experimental data apply. It was even criticized and corrected in some judicial decisions. In PRB v. Warner-Lambert Company LLC. ((2014)行提字第8号), for example, the Supreme Court articulated that for experimental evidence submitted after the filing date to prove the sufficient disclosure of the description, the evidence shall be taken into consideration if it can prove that a person skilled in the art before the filing date could carry out the claimed invention on the basis of the disclosure of the original description, and it is incorrect to totally refuse experimental evidence merely because it is submitted after the filing date. Eventually in the latest amendments to the Guidelines for Patent Examination made in 2017, SIPO revised the criteria on admissibility of late-filed experimental data, providing that for the experimental data submitted after the filing date, the examiner shall examine it, and the technical effect to be demonstrated by the experimental data shall be obtainable by a person skilled in the art from the contents disclosed in the application. Hopefully, the revision may direct all the examiners at SIPO in the right way to deal with the supplemental experimental data.
Although the latest revision to the Guidelines for Patent Examination opens a door for entering supplemental experimental data, the strict examination will be conducted on such data under the very high standard of admissibility. Actually the experimental data will be taken into consideration only when the technical effect to be demonstrated has already been asserted in the original description rather than some “new” one mentioned nowhere in the description; and the experimental date must be obtainable by a person skilled in the art from the disclosure of the original description.
All in all, the best practice for procuring patents in China is putting sufficient experimental data in the original description at the very beginning. Highly risky is to rely on late-filed supplemental experimental data to cure the deficiencies such as lack of inventiveness and insufficient disclosure.
-
ABOUT US
-
PRACTICES
-
NEWS & EVENTS
-
Other Links
-
Contact Us
- info@beijingeastip.com
- +86 10 8518 9318
- +86 10 8518 9338