With over 200 dedicated professionals, Beijing East IP has helped a full spectrum of clients – from startups to Fortune 500 corporations to domestic multinational companies – on their intellectual property issues in China.
Misrepresented Experimental Data Will Kill a Patent
On February 28, the Beijing IP Court (“IP Court”) issued a decision in Qilu Pharmaceutical v. the PRB ((2017)京73行初字No. 5365), which reversed a ruling by the Patent Reexamination Board (“PRB”) that upholds the validity of Patent No. 200910176994.1 (the “’994 patent”) owned by Beijing Sihuan Pharmaceutical (“Sihuan”). The IP Court holds that the inauthentic experimental data presented in the original description result in the failure to satisfy the enablement requirement.
Background
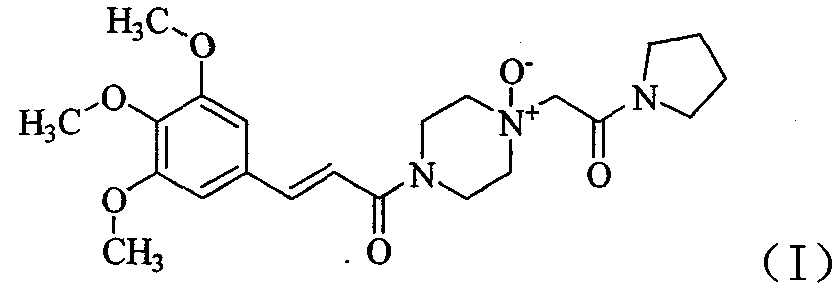
The ’994 patent claims a new cinepazide N-oxide of formula (I), a method for the preparation of the compound, and use of the compound as a standard or a reference.
Additionally, as stated in the description, it was surprisingly found that the compound has a “fairly good” insecticidal activity. Therefore, the patent also claims an insecticidal composition comprising an effective amount of the compound. Cinepazide or cinepazide maleate has been used as a first-line medication in China for the treatment of cardiovascular and cerebrovascular diseases. Granted the new drug protection and as well the relevant patents, Sihuan has been dominating the market for years.
In early 2015, Sihuan brought in different venues a series of lawsuits against Qilu Pharmaceutical (“Qilu”) for infringement of its patents directed to cinepazide, including the ’994 patent. Sihuan alleged that Qilu had infringed the ’994 patent by making the cinepazide N-oxide, and by using the compound as a standard. The lawsuits just followed Qilu’s launch of the cinepazide maleate injection into the market immediately after the expiration of the new drug protection term granted to Sihuan on the cinepazide maleate injection.
The lawsuit asserting the ’994 patent has already been concluded with a second-instance judgment favorable to Sihuan before the High Court of Inner Mongolia Autonomous Region in August 2017, awarding 1 million RMB damages for patent infringement and a post-trial injunction against Qilu.
Invalidation and Court Proceedings
On September 22, 2016, as a countermeasure against the complaint for patent infringement, Qilu filed with the PRB a petition for invalidation of the ’994 patent on the grounds of failure to meet the enablement requirement, failure to meet the support-by-the-description requirement, lack of novelty, and lack of inventiveness.
It is notable that besides the petition for invalidation, in early 2017 Qilu filed a lawsuit with the IP Court against Sihuan for monopoly and unfair competition behaviors, seeking 4 million RMB damages caused by, among others, Sihuan’s abuse of patent right that was illegally procured by presenting false experimental data.
On June 12, 2017, the PRB rendered a decision that upholds the validity of the ’994 patent.
Qilu appealed the PRB’s decision to the IP Court on the ground that, among others, the description of the ’994 patent fails to meet the enablement requirement because false experimental data were disclosed in the description to verify the insecticidal activity of the patented compound.
Looking into the description of the ’994 patent, one can find that only one working example, i.e., Example 5 “Study on Activity of Cinepazide N-oxide”, was carried out to provide experimental data concerning the insecticidal activity of the patented compound. According to the disclosure of the working example, as the “treated group”, 10 third-instar larvae of Mythimna separata were kept in a petri dish and fed with 1 cm × 1 cm corn leaves which had been soaked in an aqueous solution of cinepazide N-oxide, and as the “control group”, another 10 larvae were fed with 1 cm × 1 cm corn leaves which had been soaked in pure water. The tests were repeated three times for both the treated and control groups. As the experiment results, the antifeedant rates, the death rates, and the corrected death rates, respectively at the 24th hour and 48th hour, were reported to demonstrate the insecticidal activity of the patented compound. In particular, the antifeedant rate was reportedly 67.93% at the 24th hour and 96.57% at the 48th hour; the death rate of the treated group was reportedly 61.74% at the 24th hour and 92.71% at the 48th hour.
Qilu alleged that the experiment results of Example 5 were false according to the calculated results by using the experimental data that 1) the reported death rates of the treated group couldn’t result in integral individuals for the dead larvae, given that in total 30 larvae were subjected to the experiment in the treated group; and 2) the reported antifeedant rates would lead to the result that the average feeding amount of the control group at the 48th hour would be 9.35 times as much as that at the 24th hour, which is evidently against the normal behavior of Mythimna separata larvae.
The IP Court held that Qilu had fulfilled the primary burden of proof on challenging the authenticity of the experiment results by providing the mathematical calculation based on the given experimental data, and thus shifted the burden of proof to Sihuan. To this end, the IP Court required Sihuan to submit the original experiment record, which should have the greatest weight of proof.
Sihuan failed to submit the original experiment record, explaining that it got lost as the experiment had been carried out many years ago by its affiliated lab. Sihuan, however, defended authenticity and reliability of the experimental data. In particular, Sihuan argued that the number of Mythimna separata larvae used for the treated group was 210 in total (10 larvae × 7 dishes × 3 batches), and for the control group 60 in total (10 larvae × 2 dishes × 3 batches); the result that the feeding amount of the control group at the 48th hour was 9.35 times more than that at the 24th hour was due to the larvae’s development from the third instar to the fourth during the latter 24 hours.
The IP Court disagreed with Sihuan, holding that its arguments were inconsistent with the disclosure of the original description which now should have the greatest weight of evidence, and thus unpersuasive. Denying the authenticity of the experimental data presented in the working example, the IP Court found that the description of the ’994 patent fails to provide sufficient experiment results to verify the insecticidal activity of the patented compound. As a result, the IP Court reversed the PRB’s decision.
It is no doubt that the IP Court’s decision is a huge hit against Sihuan in contrast to its winning in the previous patent infringement lawsuits. Moreover, the IP Court’s finding on the misrepresented experimental data indicates that even if Sihuan appeals, it will have a very low (if not zero percent) chance of success. The invalidation of the ’994 patent will finally bring an end to Sihuan’s dominance of the cinepazide injection market. More critically, it’s highly possible that Sihuan will also lose the unfair competition lawsuit.
The Takeaway
Patent applicants are, of course, required to prosecute patent applications with candor, good faith, and honesty. In practice, the examiners at the Patent Office may have limited resources to examine whether experimental date presented in the patent applications are authentic or not, and therefore are difficult to capture and punish the conduct of misrepresenting experimental data in examining patent applications.
To risk misrepresenting experimental data for procuring patent right, however, is not just a game of cops and robbers between patent applicants and the Patent Office. As one can learn from the present case, patentees’ improper conducts once performed to procure their patents, such as misrepresenting experimental data in the description, will finally kill their patents. More than invalidation of patents, such improper conducts can incur severe penalties such as a counterclaim by alleged patent infringers.
-
ABOUT US
-
PRACTICES
-
NEWS & EVENTS
-
Other Links
-
Contact Us
- info@beijingeastip.com
- +86 10 8518 9318
- +86 10 8518 9338